Have you ever heard of a “frog battery”? Well, in 1845 a scientist put a strip of metal in a dead frog’s leg and an entirely different strip of metal in the other leg. And low and behold, there was an electric charge.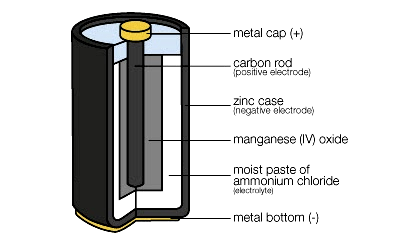 The diagram of a real battery shows the three key elements of a battery. A positive electrode (cathode), a negative electrode (anode) and the electrolyte, the muscles in the frog’s legs, or whatever material is used, across which the electrons and ions flow. Don’t worry about ions. They are members of the electron family.
The diagram of a real battery shows the three key elements of a battery. A positive electrode (cathode), a negative electrode (anode) and the electrolyte, the muscles in the frog’s legs, or whatever material is used, across which the electrons and ions flow. Don’t worry about ions. They are members of the electron family.
So, without getting techy, if you can change the composition of the electrodes and change the electrolyte, you have any number of possible batteries. And on a world scale we have something like a billion batteries in use.
The technical names are confusing, like zinc-carbon batteries, lead-acid batteries, nickel-cadmium batteries and the hot new lithium-ion batteries.
Right at my desk I am looking at different kinds of batteries. I have small batteries for my hearing aid, and they last about a week. There is a battery in a gadget I use to remove hair from my ears. How embarrassing. Ever since the hair on my head disappeared, my ear hair is trying to take over. This battery lasts about three months. And I have a small battery in an old quartz watch, and it will last a couple of years. These are very basic batteries.
Now the batteries in my smart phone, my I-Pad and my computer are in a class of their own. They are all rechargeable. These are lithium-ion batteries. They can store more electricity.
Back in my days as an engineering student, we studied the lead-acid batteries that are so important for the operation of the internal combustion engine. The power from the battery starts the engine, and the alternator on the engine charges the battery when the car is running.
The professor said, at the time, that the lead-acid battery with two different lead electrodes and a sulphur dioxide acid electrolyte was an application of the current science. And, of course, that is what engineering is about. Applied science.
Today, the secret of the electric vehicle is new lithium-ion batteries. And the auto companies like Tesla and Mercedes-Benz are bringing new batteries onto the market that hold a charge longer, can be charged more quickly and are cheaper.
It is not crazy to believe that half the automobiles in the world will be electric by 2050. Why? EVs are better technology. And, as a result, half of the oil refineries in the world will be shutting down. And, the other part of this story is the new battery technologies needed to store the power generated by solar and wind. Solar is only good when there is sunshine, and wind is only good when it is windy. The photo shows a monster battery facility outside a wind farm in Australia. Mega buzz here. Quirky.
And, the other part of this story is the new battery technologies needed to store the power generated by solar and wind. Solar is only good when there is sunshine, and wind is only good when it is windy. The photo shows a monster battery facility outside a wind farm in Australia. Mega buzz here. Quirky.
The scary part about the demand explosion for lithium-ion batteries is the metals used for the electrodes. Things like cadmium, magnesium and lithium. A Tesla battery contains, for example, 63 kg of lithium. And the big suppliers of lithium are China and Australia. But, guess what? There has been a significant discovery of lithium and other goodies in Afghanistan. Love the photo of a local with a large block of ore over his shoulder. I guess we know why the US will be staying in Afghanistan.
But, guess what? There has been a significant discovery of lithium and other goodies in Afghanistan. Love the photo of a local with a large block of ore over his shoulder. I guess we know why the US will be staying in Afghanistan.
03-03 Battery Buzz
(blank) » John Bulloch » 20 Quirky Climate » 03 Technology »
Frog batteries? Half the number of cars will be EVs. Half the refineries will shut down.